In a blog for health professionals, Cardiology Registrar Dr Ven Gee LIM reviews the latest evidence surrounding the new generation anti-diabetic agent Sodium Glucose cotransporter 2 (SGLT2) inhibitors. SGLT2 inhibitors were one of the drugs included in the 2018 Cochrane Systematic Review: Insulin and glucose-lowering agents for treating people with diabetes and chronic kidney disease by Lo et al [1].
Unexpected positive findings from the latest cardiovascular outcome trials on SGLT2 inhibitors since the 2018 Cochrane review
Since the Cochrane Systematic Review by Lo et al, several clinical trials on SGLT2 inhibitors have yielded unexpected and fascinating findings. Back in 2018, Lo et al concluded that SGLT2 inhibitors were probably efficacious for glucose-lowering and reducing blood pressure and heart failure in patients with diabetes and chronic kidney disease [1]. The SGLT2 inhibitor that was referred to by Lo et al was empagliflozin [2] but since then, two landmark SGLT2 inhibitor trials involving canagliflozin and dapagliflozin have been published [3,4] and have affirmed the hypothesis that SGLT2 inhibitors may exert cardioprotective and renoprotective effects independent of its glucose-lowering effects.
What are SGLT2 inhibitors and how did the excitement around SGLT2 inhibitors begin?
SGLT2 is a low affinity, high capacity sodium-glucose cotransporter that is expressed almost exclusively in the kidney and accounts for about 90% of renal glucose reabsorption [5,6,7]. SGLT2 inhibition reduces hyperglycaemia due to glycosuria while leading to the restoration of tubuloglomerular feedback (impaired in diabetes) and the normalisation of glomerular filtration rate (GFR) [8]. In other words, this is an anti-hyperglycaemic drug that was designed to work on the kidneys (there are no known SGLT2 receptors in the heart).
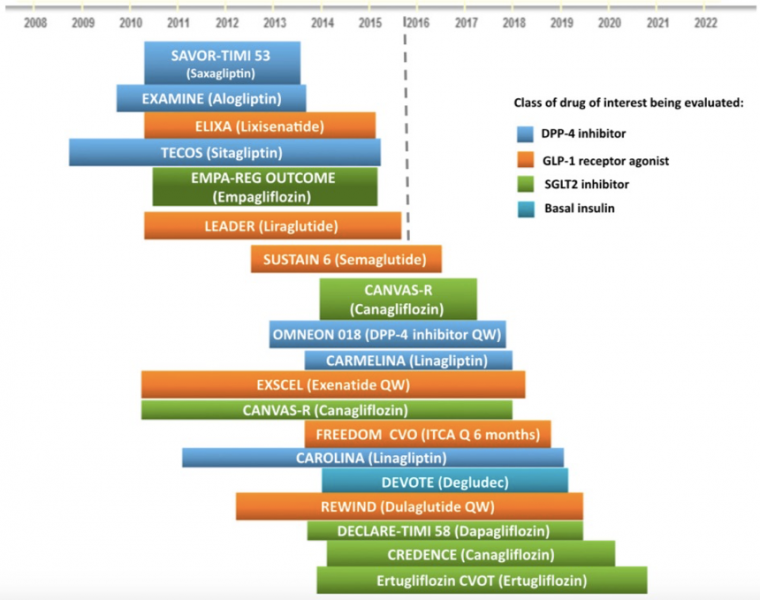
The mainstay treatment of diabetes has predominantly centered on reducing hyperglycaemia with the belief that having good glycaemic control may reduce the cardiovascular (CV) risk from diabetes. That was the basis behind the use of metformin, sulphonylureas and insulin as part of standard medical therapy. However, the trials from previous generation anti-hyperglycaemic medications on CV risk have yielded conflicting results, with the most concerning being the thiazolidinediones (rosiglitazone and pioglitazone) and their associated increase in heart failure rates [9,10]. Therefore, in 2008 the U.S Food and Drug Administration (FDA) and the European Medicines Agency (EMA) released a joint guideline calling for the demonstration of cardiovascular safety of novel anti-hyperglycaemic drugs in diabetes patients [11,12]. Since then there has been a significant growth in the number of cardiovascular outcome trials (CVOT) for the new generation anti-hyperglycamic drugs which includes the DPP-4 inhibitors, GLP-1 receptor agonists and SGLT2 inhibitors. A timeline illustrating the CVOTs for the novel anti-hyperglycaemic drugs is shown in Figure 1.
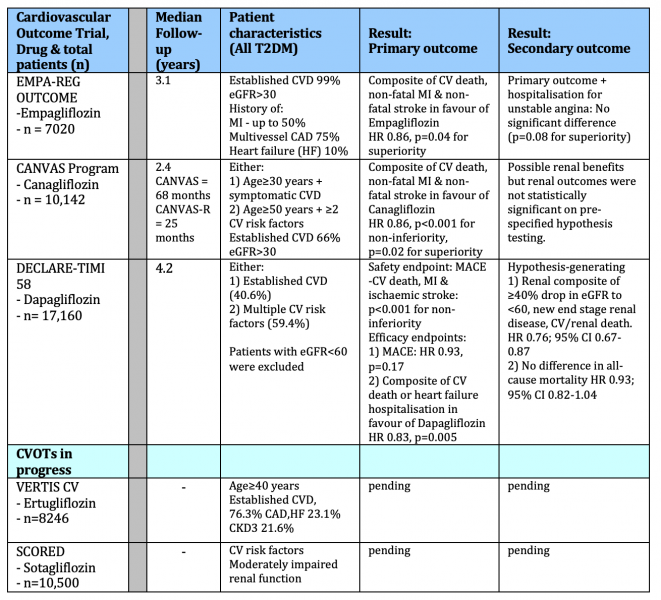
This blog will summarise the three landmark CVOTs for SGLT2 inhibitors, which all yielded fascinating results: EMPA-REG OUTCOME (Empagliflozin)[2], CANVAS Program (Canagliflozin [3] and DECLARE-TIMI 58 (Dapagliflozin) [4]. All 3 CVOTs were multicentre randomised, double-blind, placebo-controlled trials, designed to evaluate the cardiovascular safety of oral SGLT2 inhibitors in type 2 diabetes mellitus (T2DM) patients with either established cardiovascular disease (CVD) or high cardiovascular (CV) risk. All patients received a background of standard secondary prevention therapy and anti-hyperglycaemic agents.
EMPA-REG OUTCOME (Empagliflozin):
Trial design:
7,020 patients were randomised to receive either placebo or empagliflozin. This trial enrolled a patient population of high CV risk with a long duration of T2DM and established CVD: almost 50% of the patients had a history of myocardial infarction (MI), 75% had multivessel coronary artery disease and 10% had a history of heart failure. The median follow-up period was 3.1 years.
Result:
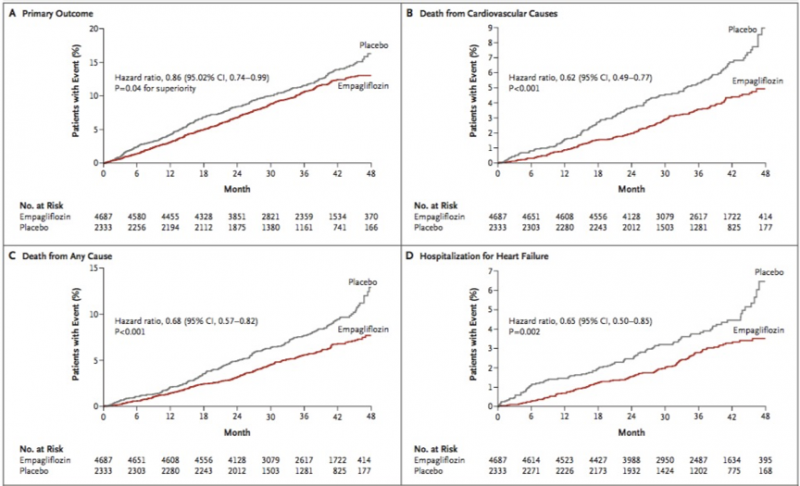
Unexpectedly, patients in the empagliflozin arm had a significant 14% reduced risk for the primary composite outcome of major adverse cardiovascular events (MACE) comprising CV death, non-fatal MI and non-fatal stroke. Specifically, the primary outcome occurred in 10.5% patients in the pooled empagliflozin group vs 12.1% in the placebo group, with a hazard ratio of 0.86 in the empagliflozin arm (95%CI 0.74-0.99; p <0.001 for non-inferiority and p = 0.04 for superiority). This reduction in MACE was driven primarily by a 38% reduction in CV death (HR 0.62; 95%CI 0.49-0.77; p <0.001). In addition, empagliflozin led to a significant 35% reduction in hospitalisation for heart failure (HR 0.65; 95%CI 0.50-0.85; p = 0.002). Finally, empagliflozin reduced the overall mortality by 32% (HR 0.68; 95%CI 0.57-0.82; p <0.001). With an absolute risk reduction of 2.6%, this translates to a number-needed-to-treat (NNT) of 39 over 3 years to prevent one death. What made the results even more fascinating was the early separation of curves, suggesting an early cardioprotective effect that may be occurring at a molecular or cellular level (see Figure 2).
CANVAS (Canagliflozin):
Trial design:
10,142 patients were randomised to receive either placebo or canagliflozin. This trial enrolled a T2DM patient population that were either: at least 30 years old with a history of symptomatic CVD or at least 50 years old with at least 2 of the following CV risk factors - hypertension, diabetes duration of at least 10 years old, current smoking, micro- or macroalbuminuria or a low HDL level (<1mmol/L). 66% of patients had a history of prior CVD (compared to 99% in EMPA-REG OUTCOME) and 14% had a history of heart failure (compared to 10% in EMPA-REG OUTCOME). The median follow-up period was 2.4 years.
Result:
Similar to EMPA-REG OUTCOME, patients in the canagliflozin arm had a significant 14% reduced risk for the primary composite outcome of major adverse cardiovascular events (MACE) with a hazard ratio of 0.86 in the canagliflozin arm (95%CI 0.75-0.97; p <0.001 for non-inferiority and p = 0.02 for superiority). This reduction in MACE was driven primarily by a 33% reduction in hospitalisation for heart failure (HR 0.67; 95%CI 0.52-0.87). Unlike EMPA-REG OUTCOME, the separation of primary outcome curves was less pronounced in the CANVAS Program (see Figure 3). Although the pre-specified hypothesis testing sequence for renal outcomes were not significant, there was a possible benefit of canagliflozin with regard to progression of albuminuria (HR 0.73; 95%CI 0.67-0.79) and composite outcome of a sustained 40% reduction in eGFR, need for renal replacement therapy or renal mortality (HR 0.60; 95%CI 0.47-0.77).
![Cardiovascular outcome in the integrated CANVAS Program. Taken from Neal et al [3] which shows the cumulative incidence of the primary composite outcome on CV mortality. The inset shows the same data on an enlarged y axis. Note that the separation of curves in the canagliflozin arm (blue curve) is less pronounced than that of EMPA-REG OUTCOME.](/sites/uk.cochrane.org/files/uploads/figure_3._ven_gee_lim_blog.png)
DECLARE-TIMI 58 (Dapagliflozin):
Trial design:
17,160 patients were randomised to receive either placebo or dapagliflozin. This trial enrolled a patient population with established CVD or multiple CV risk factors but compared to EMPA-REG OUTCOME and CANVAS, there was a high proportion of T2DM patients without evidence of CVD (more than 10,000 patients). In addition, DECLARE-TIMI 58 excluded patients with a creatinine clearance <60ml/min while EMPA-REG OUTCOME and CANVAS included all T2DM patients with an eGFR above 30ml/min/1.73m2. The primary safety outcome was major adverse cardiovascular events (MACE) and the two primary efficacy outcomes were MACE and a composite of CV death or hospitalisation for heart failure. The median follow-up period was 4.2 years.
Result:
Dapagliflozin met the pre-specified criterion for non-inferiority to placebo with respect to the primary safety outcome (p<0.001 for non-inferiority). In terms of the primary efficacy analyses, there was no significant difference in MACE (8.8% in the dapagliflozin group and 9.4% in the placebo group; HR 0.93; 95%CI 0.84-1.03; p = 0.17) but patients in the dapagliflozin arm had a significant 17% reduced risk in the composite outcome of CV death/hospitalisation for heart failure (HR 0.83; 95%CI 0.73-0.95; p = 0.005). This was driven by a 27% lower rate of hospitalisation for heart failure (HR 0.73; 95%CI 0.61-0.88).
Since there was only one out of two statistically significant outcome in the primary efficacy analyses, subsequent analyses of the additional outcomes were hypothesis-generating. The incidence of the renal composite endpoint which consisted of a reduction in eGFR, new end-stage renal failure or death from renal, or CV causes was 4.3% in the dapagliflozin group and 5.6% in the placebo group. (HR 0.76; 95%CI 0.67-0.87). There was no difference in all-cause mortality (HR 0.93; 95%CI 0.82-1.04).
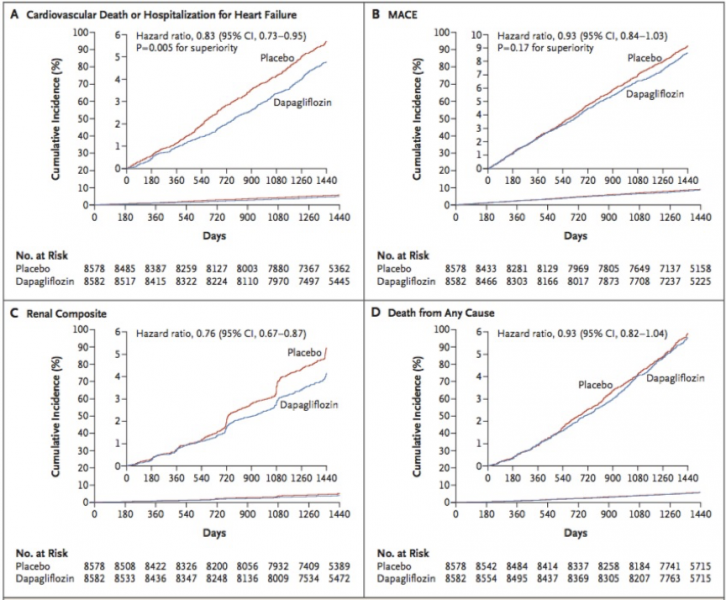
Taken from Wiviott et al [4] which shows the cumulative incidence of two primary outcomes of CV death/hospitalisation for heart failure (A), MACE (B), renal composite (C) and all-cause mortality (D). Note the early separation of curves in panel A in the dapagliflozin arm (blue curve).
What makes the findings from the SGLT2 inhibitor CVOTs so fascinating?
In summary, SGLT2 inhibition in addition to good standard of care in T2DM patients appeared to confer cardioprotection independent of blood glucose control. See Table 1 for a summary of all three landmark CVOTs and ongoing CVOTs on SGLT2 inhibitors in T2DM patients.
The main reason why the findings were unexpected and equally fascinating is that to our present knowledge, SGLT2 receptors are only found in the kidney and not the heart. The trials have all shown that an anti-diabetes drug that acts on the kidney may exert off-target effects on the heart.
What is the mechanism behind its unexpected cardioprotective effects?
The unexpected findings of the SGLT2 inhibitor CVOTs have led to an exponential increase in mechanistic studies in an attempt to elucidate the mechanism behind the pleiotrophic effects of SGLT2 inhibition. This is beyond the scope of this blog but the following is a list of several attractive ideas (in no particular order of importance):
However, there is yet to be a unifying explanation behind the pleiotrophic effects of SGLT2 inhibitors and multiple basic science and clinical studies are ongoing to help address our gap in knowledge in this field.
Limitations
It is worth remembering that SGLT2 inhibitors, like all drugs, have side effects (the most common being genitourinary infection) and have been found to be associated with adverse events such as hypoglycaemia, euglycaemic diabetic ketoacidosis, acute kidney injury, bone fracture and lower limb amputation [14]. As these are relatively new drugs, the long-term side effect profile and unanticipated drug interactions remains unknown.
Where does this leave us?
The novel findings from the SGLT2 inhibitor CVOTs has only led to more unanswered questions.
1) How does this drug work on the heart?
2) Could this drug be potentially extended to patients with end stage renal disease, type 1 diabetes or even those without diabetes?
3) Since patients with heart failure with reduced ejection fraction (HFrEF) seem to benefit from this drug regardless of the presence of diabetes (DAPA-HF trial; [15), could the same be said about an equally challenging group of patients with heart failure with preserved ejection fraction (HFpEF)?
4) Should Cardiologists now be proactive in prescribing SGLT2 inhibitors in T2DM patients or should this responsibility primarily remain with the Diabetologists? Furthermore, since the CREDENCE trial [16] showed that chronic kidney disease (CKD) patients had improved renal and cardiovascular outcomes, could Renal Physicians be potentially involved in the prescription of SGLT2 inhibitors?
Fortunately, there are multiple research groups worldwide that are performing basic science and clinical studies that will help answer some of the above questions.
Take home points
1) There are three landmark cardiovascular outcome trials of SGLT2 inhibitors that have led to plenty of excitement in the fields of cardiology and diabetes – EMPA-REG OUTCOME (empaglifozin), CANVAS (canagliflozin) and DECLARE-TIMI 58 (dapagliflozin).
2) SGLT2 inhibitors improve hyperglycaemia by acting on the kidneys but have been found to exhibit cardioprotective effects in an off-target manner.
3) The cardioprotective effects appear to occur independent of blood glucose lowering, reduction in blood pressure and weight.
References may be found here
 Ven Gee Lim @v3n_3
Ven Gee Lim @v3n_3
Ven is currently a Clinical Lecturer at the University Hospitals Coventry and Warwickshire NHS Trust and a post-doc research fellow at the University of Warwick. He completed his Cardiology training at the West Midlands Deanery with a subspecialty interest in Electrophysiology and Devices.
Ven graduated from the University of Edinburgh (Honours) in 2009, obtained his MRCP in 2012 and completed his Masters in Internal Medicine (Distinction) at his alma mater in 2014. He then pursued a PhD in Cardiovascular Science at University College London (out-of-programme research) where the focus of his research was on the role of SGLT2 inhibitors in cardioprotection and graduated in 2021.
Ven Gee Lim has no conflicts of interest to declare.
Posted: December 2019
